37 lead electron dot diagram
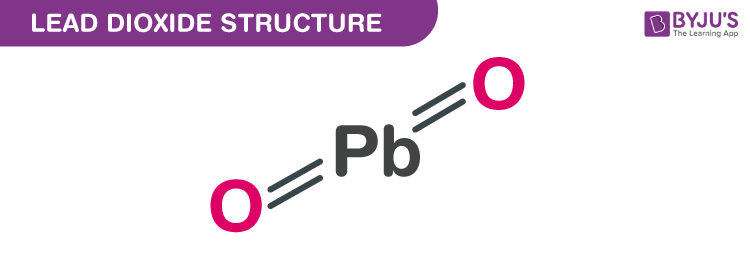
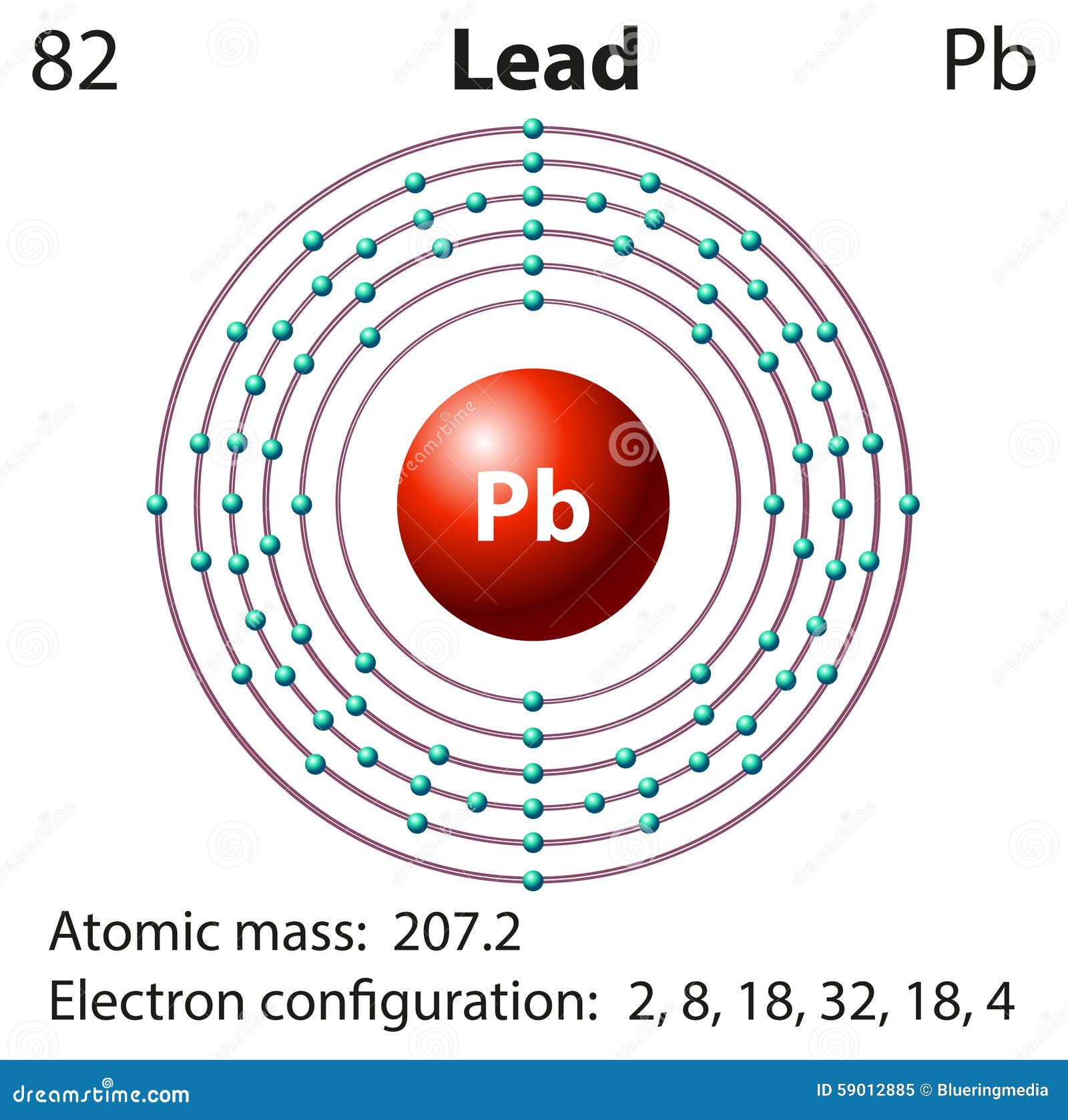
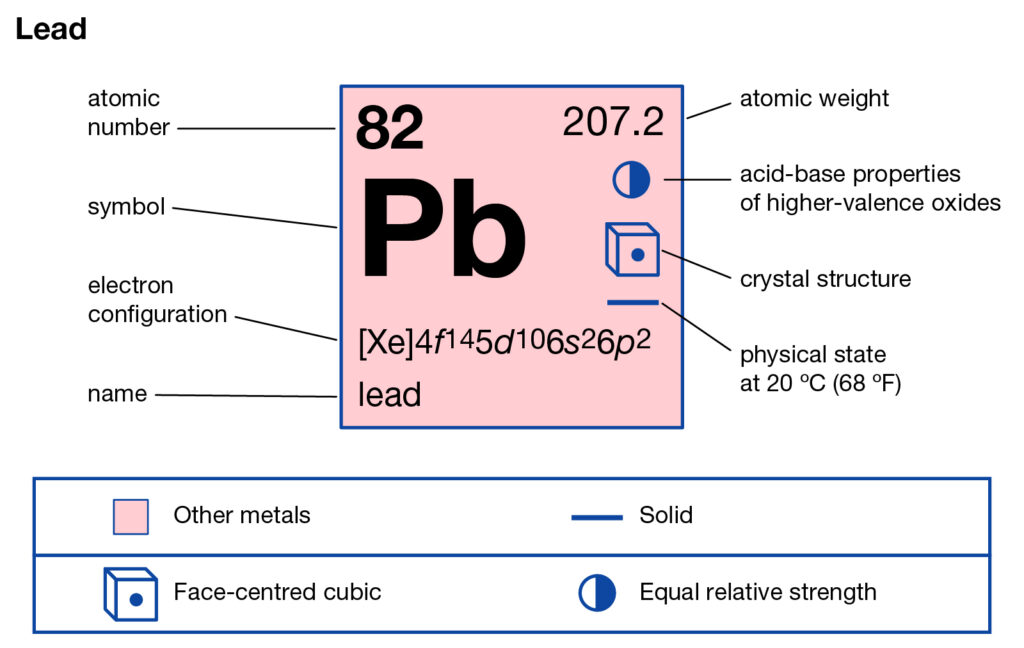
Online Posting Date: 28 April 2014. Hazardous Substances Data Bank (HSDB) Lead (IV) dioxide solids (PbO2) can form as constituents of scales of lead corrosion products that develop in lead pipes via oxidation-reduction potential induced by residual free chlorine which can effect drinking water distribution systems (1). Answer and Explanation: 1. Lead is in group 4A in the periodic table, meaning it has 4 valence electrons. That said, add 4 electrons around the symbol of lead to obtain a Lewis structure of:
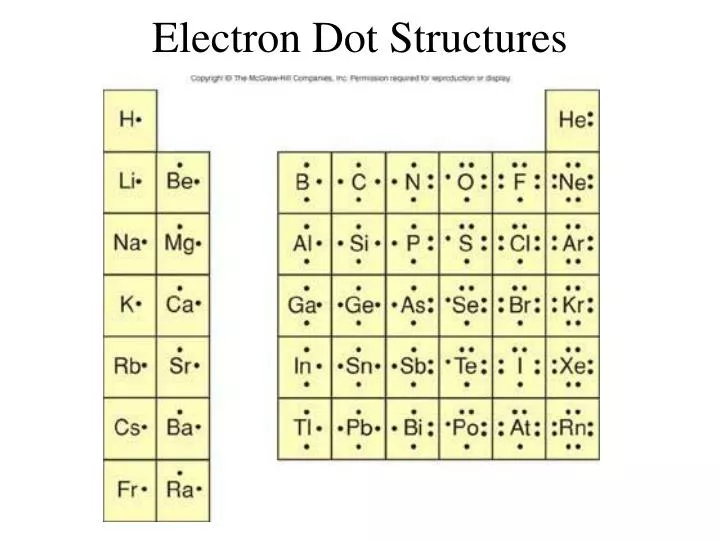
Lewis structure diagram showing lone pairs and bonding pairs of electrons in a molecule or an ion Lewis symbol symbol for an element or monatomic ion that uses a dot to represent each valence electron in the element or ion lone pair two (a pair of) valence electrons that are not used to form a covalent bond octet rule

Lead electron dot diagram
Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips with the basics. Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element's symbol. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Since electrons repel each other, the dots for a given atom are distributed evenly around the symbol before they are ...
Lead electron dot diagram. Pb with four dots around it. Since Lead (Pb) has four valence electrons (Group 14), it gets four little spots to indicate the val e- around it. Note: Make sure to not pair them up( meaning put two dots beside each other) unless you have to. The Lewis Structure uses Hund's rule when placing the dots around the element. (Only for elements with 1-4 valence electrons do you have to follow the rule ... A step-by-step explanation of how to draw the Pb(OH)2 Lewis Dot Structure.For Pb(OH)2 we have an ionic compound and we need to take that into account when we... CHEMISTRY LAB: ELECTRON DOT DIAGRAMS FOR IONIC COMPOUNDS Procedure 1) Follow the steps listed below to fill in the data table for the first 5 compounds, then on the back create a new data table for the remaining 5 compounds. 2) In drawing the electron dot diagrams use one color for the cation and a different color for the anion. A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the ...
Every chemistry student has to learn how to draw Lewis Dot Structures. The key is to understand the steps and practice. Lewis Structures are important to learn because they help us predict: the shape of a molecule. how the molecule might react with other molecules. the physical properties of the molecule (like boiling point, surface tension, etc.). Review what a Lewis dot diagram is and discover how to draw a Lewis dot structural formula for compounds. Learn how to represent single, double and triple bonds with lines instead of dots. Also ... The electron arrangement of any particular atom shows. ... Which of the following is the correct electron-dot structure for carbon? C with 4 dots. ... lead. The common unit of radioactivity which is used to measure the biological damage is the _____ rem. Lewis Structure Examples. The Lewis electron dot structures of a few molecules are illustrated in this subsection. 1. Lewis Structure of CO2. The central atom of this molecule is carbon. Oxygen contains 6 valence electrons which form 2 lone pairs. Since it is bonded to only one carbon atom, it must form a double bond.
To use Lewis electron dot symbols to predict the number of bonds an element will form. At the beginning of the 20th century, the American chemist G. N. Lewis (1875-1946) devised a system of symbols—now called Lewis electron dot symbols. A system that can be used to predict the number of bonds formed by most elements in their compounds. Skip to page content; Skip to site menu on this page. Periodic Table of Elements Element Lead - Pb. Comprehensive data on the chemical element Lead is provided on this page; including scores of properties, element names in many languages, most known nuclides of Lead. The dot diagram for lead is: Pb There are two types of diagrams one is the Lewis diagram the other is the Electron dot diagram. Lewis Structure (electron dot diagram) for the oxygen molecule, O 2, OR . There are 2 bonding pairs of electrons shared between the 2 oxygen atoms, and each oxygen atom also has 2 lone pairs (non-bonding) pairs of electrons. In the Valence structure for the oxygen molecule, each bonding pair of electrons is replaced by a dash (-) to represent a ...
Ra Ra Radium 88 Lewis (Dot) Diagram: Example 5: Radium Radium is in group 2A. Radium atoms have 2 valence electrons. Ra Ra Radium For that, we have electron shell diagrams. Here are electron shell atom diagrams for the elements, ordered by increasing atomic number. For each electron shell atom diagram, the element symbol is listed in the nucleus.

Gri 3r 1421 4 Draw Electron Dot Structure Of Carbon Dioxide And Write The Nature Of Bonding Between Carbon And Oxygen In Its Molecule Or Which Lead To The Hos Number Of Carbon
Solution. Having lost its two original valence electrons, the Lewis electron dot diagram is just Ca 2+. Ca2+. The O 2− ion has gained two electrons in its valence shell, so its Lewis electron dot diagram is as follows: Test Yourself. The valence electron configuration of thallium, whose symbol is Tl, is 6 s2 5 d10 6 p1.
I got another problem with my Chemistry Homework. I need to put the Electron Dot Structure for the following. 1) Lead Oxide 2) Potassium Sulphide 3) Sodium Nitride 4) Silver Oxide 5) Copper Chloride 6) Calcium Chloride Hope somone can help
Lead (IV) Sulfide. Molecular Formula PbS. 2. Average mass 271.330 Da. Monoisotopic mass 271.920746 Da. ChemSpider ID 23253377. - Charge. This record has not been tagged. Names.
Lead is malleable, so it can be pounded into flat sheets without breaking. How does the bonding within lead help to explain this property? ... The electron dot diagram shows the arrangement of dots without identifying the element. Which element's symbol could replace the question mark in the diagram? arsenic (As)
Lewis Dot Diagrams of Selected Elements. Lewis Symbols: Electron Configuration into Shells: Index Chemical concepts Chemistry of the Elements Periodic Table . HyperPhysics***** Quantum Physics : R Nave: Go Back: Electron Distributions Into Shells for the First Three Periods. A chemical element is identified by the number of protons in its ...
The Lewis Structure uses Hund's rule when placing the dots around the element. What does each dot represent in the Lewis diagram? Lewis dot diagrams use dots arranged around the atomic symbol to represent the electrons in the outermost energy level of an atom. Single bonds are represented by a pair of dots or one line between atoms.
Write the electron configuration for each element, then show with an electron dot diagram which elements lose and gains and electrons and finally make … sure to include the correct chemical formula for each pair. 1. Sodium + fluorine 2. Calcium + oxygen 3. Calcium + fluorine 4. Lithium + oxygen 5. Magnesium + nitrogen 6. Barium + bromine 7.
Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element's symbol. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Since electrons repel each other, the dots for a given atom are distributed evenly around the symbol before they are ...
Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips with the basics.
Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds.

A Write Two Features Of Carbon Which Lead To Form Huge Number Of Compounds Write The Nature Of Bond In Organic Compounds B Give The Formula Of Two Higher Homologues Of Ethane C Draw





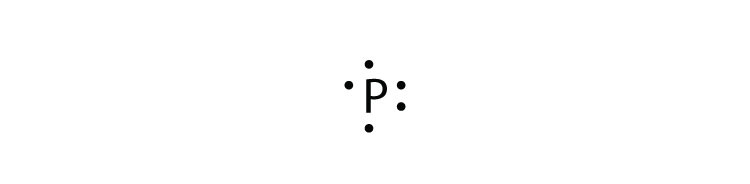















0 Response to "37 lead electron dot diagram"
Post a Comment